This web-page contains joint research with the Institute for Multiphase Processes from the Leibniz University of Hannover which was conducted within the Cluster of Excellence REBIRTH in Hannover, Germany.
Cellular response during the freeze-thaw process strongly affects the cryopreservation outcome including cell morphology and cell viability. Cryomicroscopy was used to individually analyze the osmotic response of human pulmonary microvascular endothelial cells (HPMEC) during slow cooling (1°C/min) to -60°C and fast rewarming to 4°C (100°C/min). Different concentrations of different cryoprotectant agents, dimethyl sulfoxide, ethylene glycol, proline, ectoin, and trehalose resulted in various cell volume changes. The described methods for image processing and computer vision allows for a fully automatic and individual analysis of the osmotically driven cell response under a temporal resolution of 2 frames per second. As a result, we show that in the presence of dimethyl sulfoxide or ethylene glycol cells shrink during cooling to a high degree, especially at intermediate molar concentrations in the range between 0 and 2 M, while during rewarming cells swell to isotonic volumes gradually. Comparative cell vitality tests, membrane integrity and viability tests after 24 h recultivation, under these conditions show a high cell survival. In the absence of cryoprotective agents or with proline, ectoin or trehalose, osmotic shrinkage did not meet our expectations: A sudden swelling was detected during cooling and an extreme swelling was observed after rewarming, which was accompanied by lower comparative cell viability. A linear correlation between the cellular membrane integrity after cryopreservation and the maximal relative cell volume was derived (R2 = 96). The results clearly show that it is crucial to analyse cells within a sample individually due to their individual different osmotic response.
Due to the large number of (repeated) cryomicroscopic experiments and the huge number of video frames for every sequence (each with approximately 9000 frames), there is need for a fully automatically algorithm which detects and tracks the individual cells overtime. Therefore, several (matlab)-algorithms have been developed and tested to detect and measure the volume of each cell over time. For Cell detection we applied a modified Hough-Transform
 |
 |
Cell-tracking is based on a region-growing with localized smoothness term.
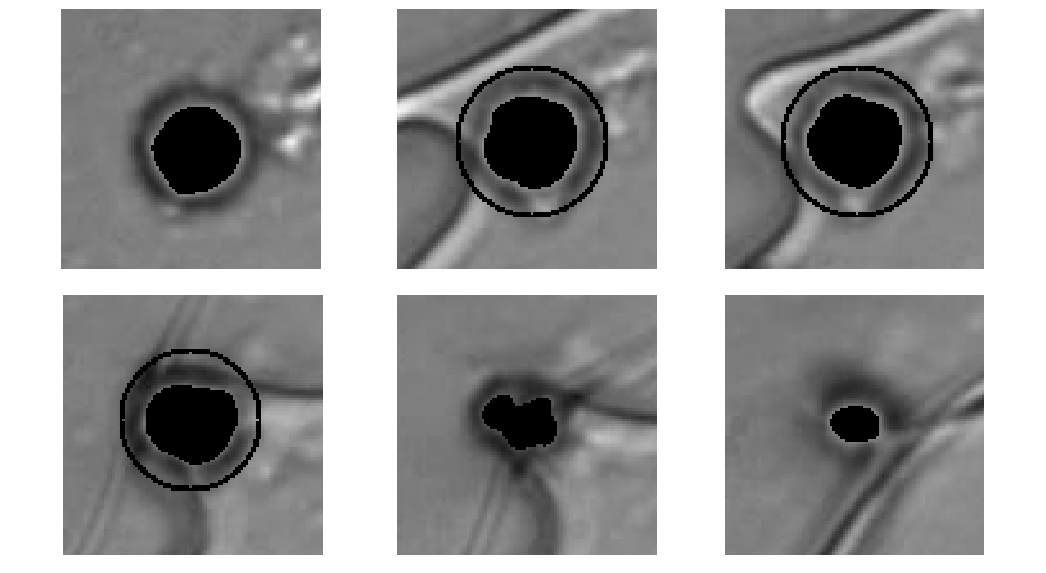
This matlab demo script gives the baseline implementation used for Celltracking in the Cryobiology paper